REIMAGINE The potential of a response
Clinical trial data support Avmapki Fakzynja Co‑Pack as a potential standard of care for adults with KRAS-mutated recurrent LGSOC1*



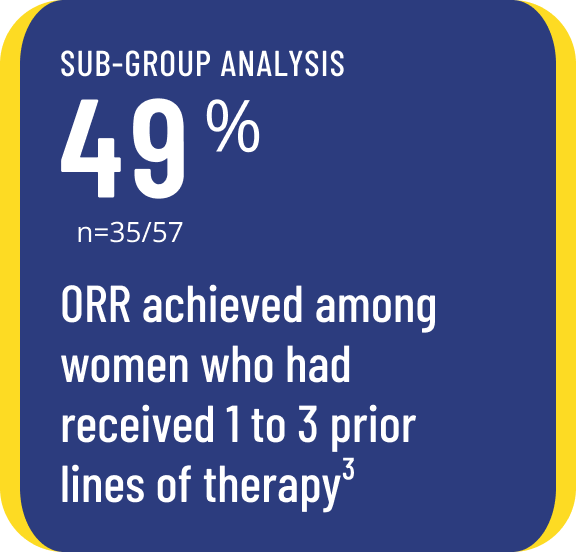




Avmapki Fakzynja Co‑Pack produced clinically meaningful response rates and DOR, with adverse events that were manageable in most patients.1
Efficacy was evaluated in RAMP-201:
an open-label, multicenter study2
Study Endpoints
• Efficacy was primarily determined by ORR as assessed by BIRC2
• Secondary endpoints included DoR, safety, and pharmacokinetics1
Patient population2
| Adult patients with measurable KRAS-mutated recurrent LGSOC | N=57 |
| Median age, years (range) | 60 (range 29-87) |
| Race and ethnicity (% of patients) |
|
| Required ≥1 prior systemic therapy, including a platinum-based regimen (lines received, % of patients) |
|
| Other prior treatments (% of patients) |
|
| Patient exclusion criteria |
|
*Who have received prior systemic therapy.
†As maintenance or treatment.
‡Including a history of retinal pathology, an active or chronic visually significant corneal disorder, or a history of glaucoma.
BIRC, blinded independent review committee; DoR, duration of response; ORR, overall response rate.
References: 1. Banerjee SN, et al. J Clin Oncol 2025;43(25):2782-2792. 2. Avmapki Fakzynja Co‑Pack. Prescribing Information. Verastem Inc; 2025. 3. Grisham RN, et al. Oral presented at Society of Gynecologic Oncology. March 14-17, 2025; Seattle, WA.